How Technology is Changing the Treatment of Type-1 Diabetes

How technology is changing the daily treatment of type-1 diabetes and impacting the lives of patients and parents.
For over 40 years, Juvenile Diabetes Research Foundation’s (JDRF) goal was to cure type 1 Diabetes (T1D). They spent years raising money for research with the one goal of ending T1D. Growing up with Type-1 Diabetes since I was 11 and having an older sister who was diagnosed with T1D when she was 6 years old, I have heard this rhetoric more than once. It has been 24 years since my sister was diagnosed (and 16 years since I was) and we still do not have a cure. However, due to technological and digital innovations, JDRF’s mission has changed. As JDRF states, “a cure is not just a destination but also a journey along a path. And we [JDRF]recognize that a part of our mission must be to help those living with T1D today to live healthier, easier, and safer lives until we arrive at the end of that path.” [1]

One of the most influential companies in changing diabetic care, and potentially JDRF’s mission, has been DexCom – a technology company founded in 1999. DexCom’s business model is to transform “diabetes care and management by providing superior continuous glucose monitoring technology” [2]. In 2006, DexCom developed its first Continuous Glucose Monitoring System (CGM) that reports glucose values every 5 minutes for 3 days through the insertion of a small sensor into your abdomen and calibrating the values every 12 hours with a manual blood glucose testing device. Prior to the introduction of the CGM, on an average day, a Type-1 Diabetic tests his / her blood sugar 8-10x and takes 4 injections of insulin (unless he is on an insulin pump). In addition, many young children who have T1D are too young to know what to do, so this responsibility lies on a parent or guardian, which can be difficult when the child is at school and the parent is at work. Now, instead of having to prick your finger to determine your blood glucose level, you can link a wearable sensor to an external device that displays your blood-sugar levels by using the technology DexCom created. The first time this technology was released, doctors and patients were guardedly optimistic as the accuracy and reliability of the device was not proven, though the potential impact of this technology on the lives of diabetics and their families could be groundbreaking.

Now ten years and several models later, through its operating model, DexCom has significantly improved its technology and offers a continuous glucose monitoring device that connects conveniently via Bluetooth to your smartphone, thereby providing greater discretion and convenience. Since the company was founded, DexCom has been dedicated to “improving the technology and helping patients…they spent $100 million in Research and Development when making the leap from the G3 [model] to the G4.” [3] Moreover, the latest DexCom model provides the option to share your blood glucose levels with a parent or guardian, which has been received positively among parents. As one father said, “It is such a change in your relationship when the first question out of your mouth when you talk to your son is no longer ‘Hey, what’s your [blood sugar] number?’ It’s ‘How was math class’?” [4] While both patients and parents are excited about the adoption of DexCom devices, the technology benefits all of the different stakeholders. For instance, insurers and doctors have also benefited as these devices have provided more reliable monitoring for doctors and reduced admissions and use of resources for hospitals, which may ultimately help lower the costs for insurance companies [5].
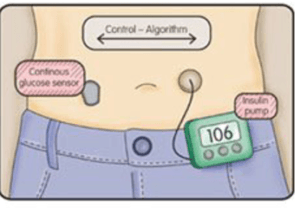
While the technology DexCom has created is important, it is also paving the way for future innovations in Diabetic care, such as a closed loop system where a CGM and an insulin pump are able to communicate directly to determine the correct dosage of insulin needed. This technology removes the human element and intuition and creates a system that runs on its own, which presents a whole new set of complications. Since blood glucose level is only one of many factors that determines one’s correct insulin dosage (other factors include exercise, stress, amount of sleep), it often seems like treating T1D is a mix of art and science because every person and every day is different. As the Vice President of Manufacturing Operations at DexCom says, “the biggest source of variability in performance is the patient” [3]. Therefore, as DexCom continues to innovate and technology becomes an even larger role in treating T1D, DexCom and other technology companies must continue to improve and perfect their existing technology before they jump to the next innovation and they must determine whether or not we can completely eliminate the human element and solely rely on science. Additionally, DexCom must continue to work with organizations like JDRF to help ensure adoption of such technology among patients and doctors.
Word Count: 805
Sources:
[1] Juvenile Diabetes Research Foundation, Mission Statement, http://www.jdrf.ca/who-we-are/mission-and-values/
[2] Dexcom, About Dexcom, http://www.dexcom.com/about-dexcom
[3] Amy Bevan, “Visiting Dexcom: An Inside View of CGM Technology”, https://myglu.org/articles/visiting-dexcom-an-inside-view-of-cgm-technology
[4] Peter Andrey Smith, “A Do-It-Yourself Revolution in Diabets Care”, http://www.nytimes.com/2016/02/23/health/a-do-it-yourself-revolution-in-diabetes-care.html?_r=0
[5] JDRF, JDRF Announces Industry Development and Discovery Parternship with Pacific Diabetes Technologies”, http://www.jdrf.org/press-releases/jdrf-announces-industry-development-and-discovery-partnership-with-pacific-diabetes-technologies/
[6] Katie Bacon, “The Future of Continuous Glucose Monitoring Technology, https://asweetlife.org/the-future-of-continuous-glucose-monitoring-technology/
[7] U.S. Food & Drug Administration, “DexCom STS Continuous Glucose Monitoring System – P050012”, http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/DeviceApprovalsandClearances/Recently-ApprovedDevices/ucm078281.htm



What a great read, and thank you so much for sharing your experiences!
As you mentioned in your article, I truly believe that the power of medical technology is in allowing patients to better monitor, understand, and control the conditions with which they live. I am impressed with the focus DexCom and the JDRF have put on making continuous glucose monitoring more available and reliable. I am curious though, about the degree to which patients and doctors are willing to trust new technology. For instance, I understand that patients with T1DM must still have their blood sugar levels intermittently checked by finger pricks in order to ensure that their continuous glucose monitors are functioning properly. Do you think that the data will one day be reliable enough to do away with this “double checking?” If so, what is the level of scrutiny that regulatory agencies need to put on these measurements, especially given that they will be used in a closed-loop control system to control insulin infusion rates. It would be a disaster if faulty glucose readings led to severe complications of hypo- or hyper-glycemia.
Great post. It’s great that the digitization of T1D treatment can both improve the quality of patients’ live and decrease costs for hospitals and insurers. It would be interesting to see if companies like DexCom focus on partnering with insurers to promote their product, as it could be to the benefit of both companies.
As far as the closed loop treatment goes, I agree with Mike that they may run into difficulties regarding approval for a system that does not rely on human oversight. Are there any other similar companies/treatments that are already doing this?
Very intersting post – thanks for sharing your personal experience dealing T1 Diabetes. I hope that you were able to benefit from this technology and am curious to hear your thoughts on its efficacy from personal experience. I think this technology is incredible and is especially useful for kids who are at school most of the day and would find it difficult to have their glucose levels monitored. I wonder if DexCom can apply this technology to other diseases that have to be monitored – to check cholesterol and blood pressure for heart disease.
I agree with Mike and Pete, it will be difficult to get the closed loop system approved by the FDA and other regulatory bodies to provide the correct dosage of insulin for patients with diabetes. It seems like there are other factors that influence insulin dosage levels other than blood glucose so I am skeptical that doctors will feel comfortable completely relying on this.