Can blockchain help solve the problem of counterfeit drugs?

Counterfeit drugs adversely impact both consumers and pharmaceutical companies. Can blockchain help solve this problem?
The problem of counterfeit drugs
Counterfeit drugs, with sales ranging from US$163 to $217 billion, are the most lucrative sector of the global trade in illegally copied goods[1], and adversely impact both consumers and pharmaceutical companies.
As for consumers, thousands of people every year die or suffer health problems after taking counterfeit drugs.[2]
From the companies’ perspective, counterfeit drugs impact their reputation, as well as damage their bottom lines. Drug manufacturers lose about US$ 205 billion annually in sales[3], and also spend large sums of money on marginally effective anti-counterfeiting measures.[4]
To tackle this issue, the U.S. Congress enacted the Drug Supply Chain Security Act (DSCSA) in 2013, which requires the pharmaceutical industry to adopt, by 2023, an “interoperable system” which will allow the FDA to identify and trace prescription drugs as they are distributed in the U.S..[5]
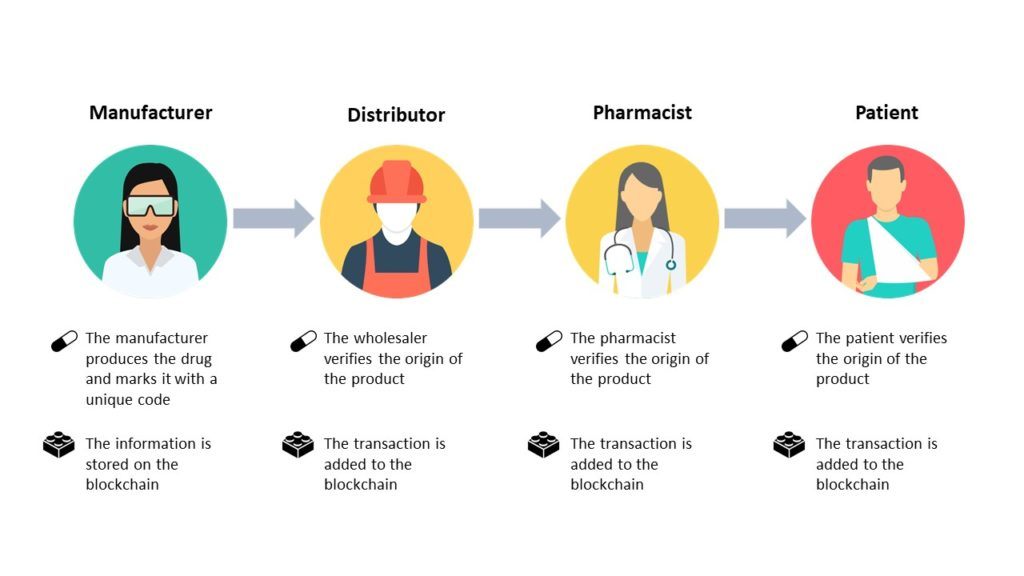
The task is far from simple: the industry will have to work together to create common standards and improve downstream communication. Manufacturers, packagers, wholesale distributors and even third-party logistics providers will play a role in creating this new system.[6]
Can blockchain help solve the problem of counterfeit drugs?
Blockchain and supply chain
In a nutshell, blockchain is a ledger, a software that and creates a tamper-proof, indelible record of transactions. This record of transactions is decentralized: every participant (node) maintains a copy of the shared ledger, and needs to approve any change. Additionally, the system is fully transparent: the list of transactions is visible to everyone.
In the context of a supply chain, blockchain can be used to track the flow of goods and services between businesses and across borders.[7] This means that, at each step of the distribution process, a network of computers can vouch for the provenance and authenticity of a shipment—making it harder for thieves to unload stolen medications, or for counterfeiters to introduce fake wares.[8]
An important advantage of using blockchain is its reliability: it is virtually impossible for malicious actors to alter the event logs. Another advantage is speed: should a shipment be disrupted or go missing, the data stored on the common ledger provides a rapid way for all parties to trace it, and determine who handled the shipment last.[9]
Pfizer’s involvement in the MediLedger project
Pfizer has teamed up with Genentech (member of the Roche group) and other pharmaceutical companies to introduce the MediLedger project, a blockchain platform to prevent counterfeit drugs from entering pharma supply chains.[10]
If the project meets its goals, everyone from drug makers to wholesalers to hospitals will be recording drug deliveries on a blockchain: its aspirations go beyond DSCSA compliance, and the participants hope to use their system to help fundamentally move the industry forward in improving drug security and preventing the production and trafficking of counterfeit drugs globally.
While is it promising that major pharmaceutical players are involved in the project, introducing a blockchain system – in which each participant controls a node in the network – will be challenging in the pharma industry, where large conservative companies dominate.[12]
The road forward
If the project succeeds, it will undoubtedly disrupt the pharmaceutical industry. However, some additional issues about blockchain will also need to be considered:
- How can blockchain address potential errors, fraudulent transactions or bugs in the code without creating data vulnerability?[13]
- Despite blockchain’s current decentralization, private ledgers will inevitably arise, as companies seek to protect market share and profits. How can interoperability across private and public blockchains be assured?[14]
Word count: 799
[1] Peter Behner et al., “Fighting counterfeit pharmaceuticals: New defenses for an underestimated – and growing – menace”, Strategy&, June 29, 2017, https://www.strategyand.pwc.com/reports/counterfeit-pharmaceuticals, accessed November 2017.
[2] Interpol, “Pharmaceutical Crime – The Dangers”, https://www.interpol.int/Crime-areas/Pharmaceutical-crime/The-dangers, accessed November 2017.
[3] Peter Behner et al., “Fighting counterfeit pharmaceuticals”.
[4] See, for instance, Jeff Swiatek, “Lilly spends millions fighting fake pharmaceuticals”, USA Today, April 6, 2013, https://www.usatoday.com/story/news/nation/2014/04/06/drugmaker-spends-millions-fighting-fake-pharmaceuticals/7357799/, accessed November 2017.
[5] U.S. F.D.A., “Drug Supply Chain Security Act”, https://www.fda.gov/Drugs/DrugSafety/DrugIntegrityandSupplyChainSecurity/DrugSupplyChainSecurityAct/, accessed November 2017.
[6] Edwin Lopez, “Big Pharma builds blockchain prototype to stop counterfeits”, Supply Chain Dive, September 21, 2017, https://www.supplychaindive.com/news/big-pharma-blockchain-MediLedger-DSCSA-FDA/505563/, accessed November 2017.
[7] Michael J. Casey and Pindar Wong, “Global supply chains are about to get better, thanks to blockchain”, Harvard Business Review, March 13, 2017, https://hbr.org/2017/03/global-supply-chains-are-about-to-get-better-thanks-to-blockchain, accessed November 2017.
[8] Jeff John Roberts, “Big Pharma Turns to Blockchain to Track Meds”, Fortune, September 21, 2017, http://fortune.com/2017/09/21/pharma-blockchain/?iid=sr-link2, accessed November 2017.
[9] Ibid.
[10] Joseph Young, “Pharma Giants Use Ethereum Network to Prevent Counterfeit Medicine”, BTCMANAGER, September 25, 2017, https://btcmanager.com/pharma-giants-use-ethereum-network-prevent-counterfeit-medicine/, accessed November 2017.
[11] Images from https://www.flaticon.com/, by authors Roundicons, Vectors Market, Icon Pond and Vectorgraphit.
[12] Jeff John Roberts, “Big Pharma Turns to Blockchain to Track Meds”, Fortune.
[13] Richard Lumb et al., “Editing the uneditable – Why distributed ledger technology must adapt to an imperfect world”, Accenture, 2016, https://www.accenture.com/t20160927T033514Z__w__/us-en/_acnmedia/PDF-33/Accenture-Editing-Uneditable-Blockchain.pdf#zoom=50, accessed November 2017.
[14] Michael J. Casey and Pindar Wong, “Global supply chains are about to get better, thanks to blockchain”, Harvard Business Review.



It’s no secret that the pharma industry is in need of some major upgrades. One glaring problem is the $220 billion worth of counterfeit drugs that plague the market.
The remaining fundamental questions executives want addressed are:
(1) who will pay for the technology
(2) who owns the data, and
(3) how companies will obtain internal and external buy-in from each of the necessary partners in the supply chain.
While the technology will likely be part of the health-care ecosystem, I wonder to what extent and how quickly it will be implemented. But I do agree that there is more promise on blockchain’s use in drug distribution than in other areas of healthcare because the supply chain does not involve individual patient protected health information.
How is the drug marked with a unique code? Is it on the packing, or printed directly on the drug, or is it just the formula that’s identified? Tying physical objects to the blockchain is a serious challenge, because any kind of sensors are subject to tampering and fraud, and blockchain’s immutability can actually be a downside (as you indicated in the error adjustment part of your ending questions).
Definitely a thought-provoking new idea for an established problem – counterfeit drugs have the gravest impact on the consumers themselves. But it is because consumers respond to emails that market them that the counterfeiters have any market to operate in at all. So I think another question would be, even if you could meet the technology and investment hurdles of implementing such a system for supply chain tracking, how do we get end consumers bought in to its credibility and importance? For example, elderly consumers who may more easily fall prey to counterfeiters may be suspicious or simply not understand the relevance of such a sophisticated tracking system, thus blunting the impact of such protection.
This article raises a really interesting point and believe there are many ways technology/digitization can be leveraged to improve the supply side of healthcare. One alternative to this approach would be equipping consumers with the tools themselves to both a) confirm they are taking the right drugs and b) conduct tests that otherwise would be required to go through the lab. Such a handful device at home could be a powerful thing: (1) The first pill out of every bottle would be ran through an assay to confirm it was the right medication; and (2) blood tests typically reserved for the lab could be conducted at home (with the appropriate assay slide) and a diagnosis could be made remotely (and even a prescription and order of the drugs could be executed). This would create a much more efficient supply chain overall for the pharma and healthcare industries (not to mention, increase patient convenience and reduce a significant amount of wasted spend to labs today).
This article is interesting in questioning
Would it be necessary to use the blockchain? To what extent does this work in the delivery of a physical product, e.g. how would the blockchain update? To what extent could products be stamped with codes identifying their batches, which are searchable on an online website?
I think that the blockchain presents a unique opportunity to track product and financial transactions in a novel manner. In other cases, it seems like it makes for a more hip approach to updating old database systems, sometimes when a traditional database system would be of equal utility with an easier implementation. The technology is in it’s infancy so it’s hard to predict – to what extent do you think it will be applied?
I imagine that delivery is a binary system, via which drug manufacturers could talk to one another. ?
This article is interesting in questioning the extent to which the blockchain can enter our daily lives. When I hear these questions, as a luddite I always ask would it be necessary to use the blockchain? To what extent does this work in the delivery of a physical product, e.g. how would the blockchain update? To what extent could products be stamped with codes identifying their batches, which are searchable on an online website?
I think that the blockchain presents a unique opportunity to track product and financial transactions in a novel manner. In other cases, it seems like it makes for a more hip approach to updating old database systems, sometimes when a traditional database system would be of equal utility with an easier implementation. The technology is in it’s infancy so I’d be hard-pressed to say it’s impossible. It will be interesting to see how it’s applied.
Blockchain clearly addresses a major issue in the pharmaceutical industry: via normal visual inspection, it is often impossible to determine whether a drug is valid or counterfeit. Blockchain adds visibility and transparency at every step of the process.
However, I feel that some members of the supply chain will be more willing than other members to participate in this blockchain management system. I imagine that implementing this system would come at significant cost to distributors, for example, who might be working with tight margins. Will distributors be able to afford and manage the technology and labor required to manage the blockchain? Additionally, it might be challenging for distributors to manage drugs that are participating in this blockchain system alongside drugs or other products that are not participating in the system, as well as keep up with any changes in the blockchain technology.